Role of Mitochondria in Metabolic Syndrome
Metabolic syndrome, previously also referred to as syndrome X or insulin resistance syndrome, is a pathological condition seen in around 20-30% of the world population and up to one in three adults in the US.
As obesity continues to rise over the years, so does metabolic syndrome. It is seen more frequently with increasing age and lower income. However, it still poses a sizable risk during childhood and adolescence, with prevalence rates of 3% and 5%. Women run a slightly higher risk of developing metabolic syndrome in certain ethnic groups, although it overall involves both gender groups equally.
In this article, let’s understand the fundamentals of metabolic syndrome at the mitochondrial level.
Diagnosing metabolic syndrome
The diagnosis is based on the following group of conditions:
- High blood pressure: systolic blood pressure of ≥ 130 mmHg, diastolic blood pressure of ≥ 85 mmHg
- Elevated blood sugar: fasting blood glucose of ≥ 100 mg/dL
- Excess fat around the waist: waist circumference of > 40 inches in men and > 35 inches in women
- Abnormal cholesterol levels: H-LDL of < 40 mg/dL in men or H-LDL < 50 mg/dL in women
- Abnormal triglyceride levels: triglycerides of ≥ 150 mg/dL
Excess fat around the waist can also be assessed by using the body shape as an indicator:
- People with apple-shaped bodies, characterized by a larger waist, may have a higher risk of developing metabolic syndrome
- People with pear-shaped bodies are not known to have a higher risk despite having a tendency to carry fat around the hips.
Metabolic syndrome is, unfortunately, far from a harmless condition. It poses a serious threat to multiple organ systems, including a higher risk of developing type 2 diabetes, heart and vascular disease, dementia, and fatty liver disease. The longer the risks, as mentioned earlier, go unmanaged, the more damage they can cause to different vital organs.
A useful watch is Dr. Robert Lustig’s presentation, titled, The New Energy Crisis, which sets out a unifying hypothesis connecting mitochondrial dysfunction to chronic disease through reactive oxygen species (ROS), linking obesogens, neurodegeneration, and metabolic disorders.
At first glance, metabolic syndrome might seem like a surface-level issue. Still, there’s a small but essential organelle that plays a deeper role: the powerhouse of the cell, namely the mitochondria.
Key role of mitochondria in metabolic syndrome
Mitochondria are crucial organelles responsible for continuously producing the energy our bodies need to function through a process called oxidative phosphorylation. What makes them particularly unique is that they possess their own DNA, distinct from the nuclear DNA found in the rest of the cell.
There are four main ways in which mitochondria have been shown to influence metabolic syndrome:
- Reduced mitochondrial DNA copy number
- Mitochondrial DNA mutations
- Proteins encoded by mitochondrial DNA
- Mitochondrial dynamics setting the stage for metabolic syndrome
Each of these is discussed in the following sections.
Reduced mitochondrial DNA copy number
In healthy cells, there are hundreds to thousands of copies of mitochondrial DNA, although these copy numbers vary across tissues and organs. Mitochondrial DNA copy numbers are reduced in patients with metabolic syndrome, especially in fat tissue, skeletal muscles, and blood cells.
This reduction in mitochondrial DNA is correlated with increased insulin resistance, higher triglyceride levels, lower HDL cholesterol levels, and a greater risk of type 2 diabetes.
We can relate this to the fact that reduced mitochondrial copies impair energy metabolism, contributing to metabolic disturbances seen in metabolic syndrome.
Mitochondrial DNA mutations
Mitochondrial DNA, unlike nuclear DNA, lacks protective packaging by histones and is therefore less likely to be able to repair itself well. This leads to a higher frequency of mutations. Such mutations can result in developing hormonal dysfunctions such as diabetes, obesity, and growth hormone deficiencies.
Scientists have been able to pinpoint specific mutations that lead to diseases, such as tRNA-Leu 3243A>G that can cause mitochondrial diabetes, often called MIDD (Maternally Inherited Diabetes and Deafness).
The same mutation can sometimes also cause MELAS syndrome, characterised by neurological problems or muscular issues. These mutations are inherited from mother to child and the severity can vary even within the same family, depending on how many mitochondria carry the mutation.
Proteins encoded by mitochondrial DNA
Mitochondrial DNA is structured differently from nuclear DNA. It’s circular and encodes a significantly lower amount of important proteins. It carries just 13 protein-coding genes, which are essential for the production of energy through oxidative phosphorylation.
These proteins are spread across several complexes of the OXPHOS system. If a mutation is to occur in these genes, the mitochondria are therefore unable to properly burn sugar and fat to produce energy.
When this fails, it can trigger a chain reaction with less energy, more inflammation, and ultimately the features of metabolic syndrome, fueling diabetes and increasing the risk of heart disease.
Mitochondrial dynamics setting the stage for metabolic disease
Mitochondria aren’t static organelles; they are highly dynamic. They constantly change shape, move, join together, split apart, make new copies, or get recycled when damaged through a process called mitophagy.
Mitophagy, in combination with mitochondria joining together, ensures a certain quality control of the cell, removing faulty mitochondria before they are able to cause harm.
Only when 60–90% of mitochondria carry the mutation does the cell get harmed. Mitochondria joining together can buffer this critical threshold by mixing healthy DNA with mutated DNA.
Intuitively, mitochondria splitting apart can then accelerate the accumulation of mutant mitochondrial DNA, leading to dysfunction. This delicate balance can be disrupted by poor lifestyle factors, which can set the stage for metabolic diseases.
Impact of mitochondrial dysfunction on the heart
Mitochondria are essential for the heart because it requires a vast amount of energy to beat continuously. In metabolic diseases, the mitochondria face multiple stressors.
Around 90% of energy production in the heart relies on oxidative phosphorylation by the mitochondria and, therefore, it only makes sense that any disruption in mitochondrial function can quickly affect cardiac performance.
The heart normally relies on fatty acids and sugar to maintain its continuous energy production. The mitochondria switch between these two options depending on their current energy needs.
In metabolic syndrome, where the cells have become more resistant to insulin, the use of sugar to produce energy is reduced. This pushes the heart to rely excessively on fatty acids.
Such a shift is, however, inefficient as it increases oxygen demand, lowers ATP yield, and generates more reactive oxygen species (ROS), causing stress to the cells.
In healthy individuals, the heart cells generate energy by burning fatty acids and sugar in the mitochondria. In diabetic patients, however, where the body has grown resistant to insulin, with high blood sugar, the body relies heavily on fatty acids. This shift decreases the efficiency and overworks mitochondria.
Mitochondrial dynamics become imbalanced
Building on the points discussed in the previous section, mitochondria are able to maintain their health thanks to their dynamic existence. In metabolic disease, we see increased splitting of mitochondria, and the proteins responsible for joining mitochondria are produced in smaller amounts. This imbalance in mitochondrial dynamics leads to fragmented mitochondria, which are less efficient at energy production.
Heart cells require precise calcium regulation in order to work effectively. Mitochondria normally take up calcium during each heartbeat to stimulate energy production in sync with each contraction. In metabolic syndrome, the mitochondria become flooded with calcium, and over time, this leads to chronic calcium mishandling, which contributes to contractile dysfunction and arrhythmias.
Overproduction of reactive oxygen species (ROS) is another significant mechanism linking mitochondrial dysfunction to heart disease. ROS contribute to cell signaling in trace amounts, while long-term overproduction damages mitochondrial DNA.
This lowers energy production efficiency and starts a vicious cycle where damaged mitochondria produce more ROS, which exacerbates the dysfunction. This eventually leads to inflammation and cardiac muscle structural remodeling.
There are two main protective systems that the mitochondria use:
- Mitophagy to remove defective mitochondria
- Biogenesis to stimulate the production of new mitochondria.
In metabolic syndrome, this protective renewal system is impaired.
The dysfunctional mitochondria accumulate as they can’t be effectively eliminated, while fewer healthy mitochondria are made. This results in an energy-starved heart that gets pushed toward failure.
Diabetic cardiomyopathy is the result of the aforementioned mechanisms together. Early on, this only manifests as minor issues with the heart’s capacity to relax and fill. But, as mitochondrial dysfunction worsens, heart muscle fibrosis and thickening occur along with contractile dysfunction.
Furthermore, diabetic cardiomyopathy can develop without high blood pressure or blocked coronary arteries, demonstrating the crucial role mitochondria play in the development of this condition.
Mitochondrial dysfunction in insulin resistance
Mitochondria oxidize fatty acids and glucose to generate energy. When mitochondria falter, fatty acids aren’t efficiently oxidized, and intermediates (i.e., ceramide and diacylglycerol) start building up in muscle cells.
Insulin resistance can result from these intermediates’ direct interference with important insulin pathways. Mitochondria must work harder to digest glucose and fatty acids when cells are overwhelmed with them, as in type 2 diabetes and obesity. As a result, the mitochondria produce more ROS, which are normally mopped up by antioxidants, but in the case of overload, start to cause stress to the cell as they are not efficiently cleaned up anymore.
The damaged mitochondria, owing to this, are then removed by mitophagy, which prevents more damage from ROS, but also reduces the overall mitochondrial population.
Because of its large mitochondrial reserve, skeletal muscle has extra capacity in the event of mitochondrial dysfunction. This implies that skeletal muscles can still meet their basic energy needs even if some mitochondria become less effective. Although it would seem that muscles could make up for the body’s ongoing nutritional excess, these little inefficiencies add up over time and cause toxic substrates to build up.
Over time, these byproducts may then directly disrupt insulin’s effectiveness, thereby shifting the system gradually in the direction of insulin resistance.
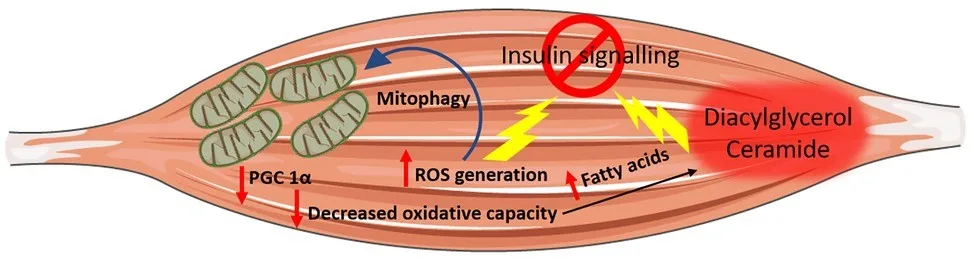
Image Credit: Sergi D, Naumovski N, Heilbronn LK, Abeywardena M, O’Callaghan N, Lionetti L, Luscombe-Marsh N. Mitochondrial (Dys)function and Insulin Resistance: From Pathophysiological Molecular Mechanisms to the Impact of Diet. Front Physiol. 2019 May 3;10:532. doi: 10.3389/fphys.2019.00532. PMID: 31130874; PMCID: PMC6510277.
Connection between mitochondrial dysfunction and insulin resistance
Scientists recently demonstrated the connection between mitochondrial dysfunction and insulin resistance by studying patients with type 2 diabetes and their first-degree relatives.
These individuals frequently displayed a reduced ability of muscle mitochondria to carry out oxidation. Additionally, they expressed fewer genes that are controlled by PGC-1α, a crucial factor that governs the creation and growth of new mitochondria. However, it is still up for debate whether mitochondrial dysfunction is a cause, a consequence, or both of insulin resistance.
Some studies have shown that mitochondrial dysfunction may precede insulin resistance, other studies have found no mitochondrial impairment in patients with insulin resistance or even observed a compensatory increase in mitochondrial capacity. Whether mitochondrial dysfunction is the main cause or a consequence of insulin resistance, improving mitochondrial function almost always improves the effectiveness of insulin.
Treating metabolic syndrome
Making regular lifestyle adjustments that address the underlying causes of metabolic syndrome is the most efficient way to combat it.
The cornerstone of this approach is adopting a healthy diet that lowers cardiovascular risk, such as the Dietary Approaches to Stop Hypertension (DASH) diet. Calorie restriction without malnutrition can also be helpful.
Exercise is the strongest booster; however, certain nutrients and bioactive compounds. such as, polyphenols and omega-3 fatty acids, can also encourage the mitochondria to function better.
Reaching and maintaining a healthy weight has a profound effect, even with about 5-10% weight loss. Weight reduction can significantly enhance insulin sensitivity and reduce cardiovascular risk.
Professional guidance from dietitians and physiotherapists, alongside all of these changes, can make the treatment way more effective and sustainable.
Read / Watch Next
- Read an article featuring a conversation with Dr. Richard Johnson, the discoverer of the Fat Switch and its connection with mitochondria
- Listen to a podcast discussion on mitochondrial function and energy resilience in aging populations
References
- Jasvinder Singh Bhatti, Gurjit Kaur Bhatti, P. Hemachandra Reddy, Mitochondrial dysfunction and oxidative stress in metabolic disorders — A step towards mitochondria based therapeutic strategies, Biochimica et Biophysica Acta (BBA) – Molecular Basis of Disease, Volume 1863, Issue 5, 2017, Pages 1066-1077, ISSN 0925-4439.
- Liang X, Or B, Tsoi MF, Cheung CL, Cheung BMY. Prevalence of metabolic syndrome in the United States National Health and Nutrition Examination Survey 2011-18. Postgrad Med J. 2023 Aug 22;99(1175):985-992. doi: 10.1093/postmj/qgad008. PMID: 36906842.
- Rus M, Crisan S, Andronie-Cioara FL, Indries M, Marian P, Pobirci OL, Ardelean AI. Prevalence and Risk Factors of Metabolic Syndrome: A Prospective Study on Cardiovascular Health. Medicina (Kaunas). 2023 Sep 25;59(10):1711. doi: 10.3390/medicina59101711. PMID: 37893429; PMCID: PMC10608643.